FDA Approves CareTaker Wireless Remote Patient Monitor

FDA Approves CareTaker Wireless Remote Patient Monitor
May 12, 2016 — CareTaker Medical announced that the U.S. Food and Drug Administration (FDA) has issued 510(k) clearance for the company's Wireless Continuous Non-Invasive "Beat-by-Beat" Blood Pressure ("cNIBP") and Heart Rate Monitor. The device is based on patented Finger Cuff technology. The wearable CareTaker monitor enables uninterrupted wire-free and electrode-free vital signs monitoring throughout the full mobile continuum of care; within the clinic and hospital, during patient transport and remotely after patient discharge. Using a comfortable, low-pressure finger cuff, CareTaker's patented Pulse Decomposition Analysis technology non-invasively measures continuous beat-by-beat blood pressure with accuracy exceeding Association for the Advancement of Medical Instrumentation (AAMI) requirements. The device also measures heart rate as accurately as a three-lead electrocardiogram (ECG) for remote display on the CareTaker secure Web portal or other wireless devices. Historically, most continuous beat-by-beat blood pressure measurements require an invasive arterial catheter or complex equipment, and have therefore been used mainly in critical care settings. The CareTaker cNIBP technology delivers ICU-quality continuous measurements without intrusive catheters or cumbersome wires, giving clinicians the ability to identify hemodynamic deterioration trends early, allowing more time for intervention while providing a comprehensive view of a patient's hemodynamic status. CareTaker's comfortable finger cuff is gentle enough to be worn continuously, even during sleep, and usage is intuitive enough to ease the clinician's workflow. The system’s onboard cellular and Bluetooth communication capabilities allow for simple setup and deployment as well as seamless integration with other devices, remote patient monitoring platforms and hospital electronic medical record systems. "CareTaker is a real game changer, allowing physicians to remotely monitor medical-grade continuous blood pressure and heart rate from anywhere, using only a patient friendly-finger cuff," said Jay Sanders, M.D., professor of medicine (adjunct) at Johns Hopkins School of Medicine and president emeritus of the American Telemedicine Association. "Until now, most clinicians have had to settle for intermittent 'point-in-time' blood pressure measurements using bulky arm cuffs, which can produce misleading results due to the influence of many factors such as movement, posture, anxiety or caffeine. In remote monitoring settings, the ability to gather continuous blood pressure and vital sign data from such an integrated, easy-to-use device will provide better information and improve patient compliance while reducing cost and workload." For more information: www.caretakermedical.net
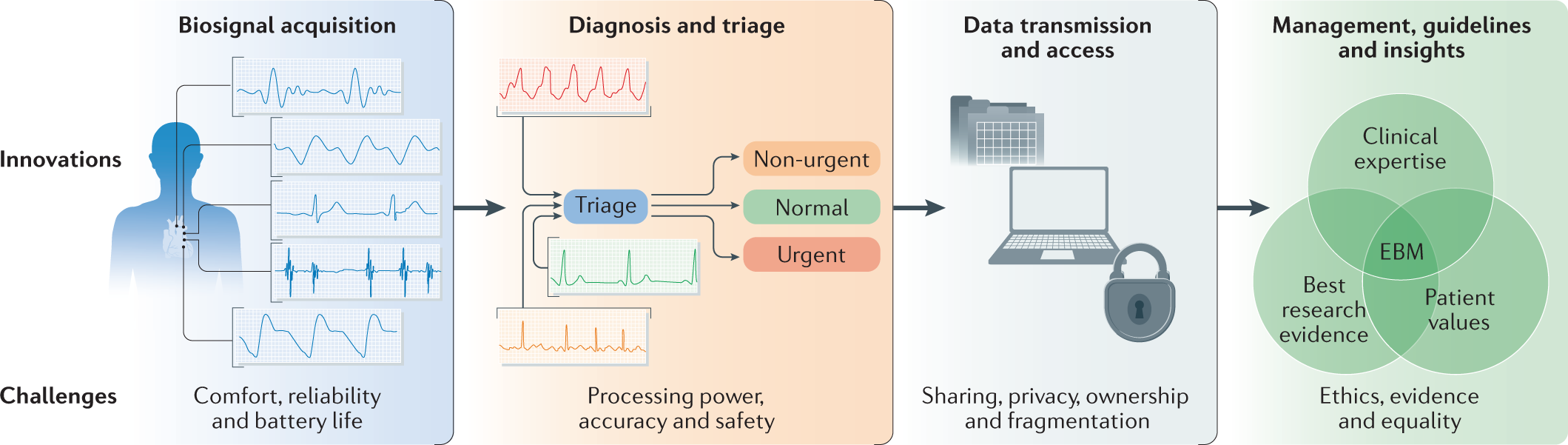
Integration of novel monitoring devices with machine learning

Patient Monitoring — Neoteric Healthcare
Custom Remote Patient Monitoring: All-Around Overview 🕹️👩🏻⚕️

LATITUDE™ NXT Remote Patient Management System – Boston Scientific

FDA roundup: The major device, app, and algorithm approvals of

Remote Patient Monitoring News and Resources for Healthcare

Telehealth and Remote Patient Monitoring for Long-Term and Post
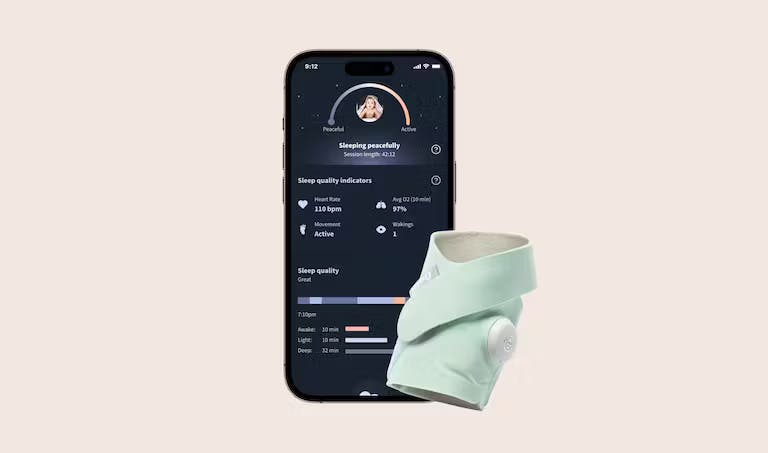
The FDA's newest approved device: A baby sock
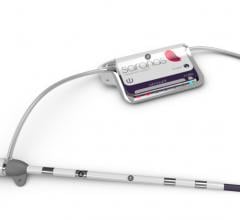
FDA Approves CareTaker Wireless Remote Patient Monitor

20/20 vision - Bluetooth wireless and medical devices past
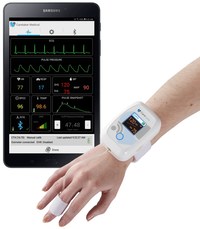
Australia's First 'Virtual Hospital' Implements Caretaker

FDA clears wireless, blood pressure-monitoring finger sensor from

FDA grants new pediatric indication for Medtronic Linq II


